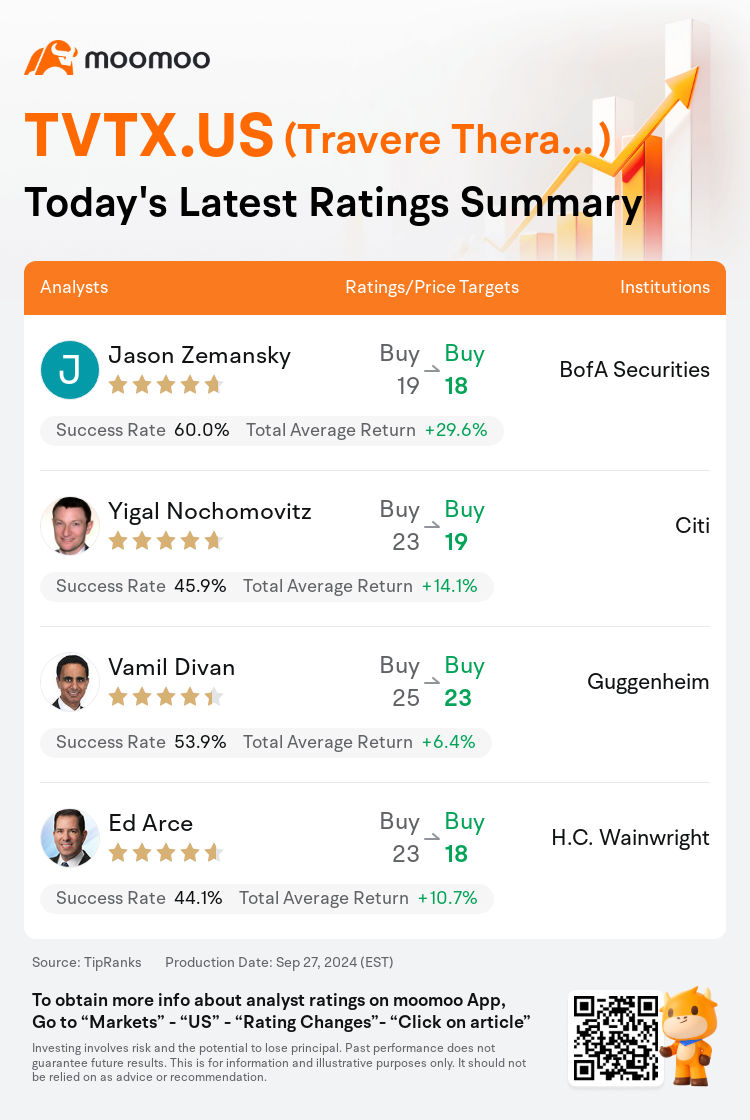
On Sep 27, major Wall Street analysts update their ratings for $Travere Therapeutic (TVTX.US)$, with price targets ranging from $18 to $23.
BofA Securities analyst Jason Zemansky maintains with a buy rating, and adjusts the target price from $19 to $18.
Citi analyst Yigal Nochomovitz maintains with a buy rating, and adjusts the target price from $23 to $19.
 Guggenheim analyst Vamil Divan maintains with a buy rating, and adjusts the target price from $25 to $23.
Guggenheim analyst Vamil Divan maintains with a buy rating, and adjusts the target price from $25 to $23.
H.C. Wainwright analyst Ed Arce maintains with a buy rating, and adjusts the target price from $23 to $18.
Furthermore, according to the comprehensive report, the opinions of $Travere Therapeutic (TVTX.US)$'s main analysts recently are as follows:
Travere Therapeutics recently announced a halt to the enrollment of their pegtibatinase phase 3 HARMONY study due to manufacturing concerns. This development is seen as a delay to the commercial portfolio, with the company anticipating a possible resumption of the trial by 2026 at the earliest. Despite the setback, the value ascribed to the program was already minimal, and the company's management has conveyed that the halt is unlikely to affect the trial's design and enrollment. Consequently, the anticipated impact on market sentiment in the near term is expected to be limited.
Travere Therapeutics announced a voluntary suspension in the enrollment for their Phase 3 HARMONY study of pegtibatinase, attributed to challenges in manufacturing scale-up. This development is seen as a setback given that pegtibatinase was anticipated to expand the company's portfolio beyond Filspari. The anticipated market introduction has now been adjusted to fiscal 2030, with the assumption that enrollment will recommence in 2026.
After Travere Therapeutics announced a voluntary pause in the Phase 3 HARMONY study for pegtibatinase in the treatment of classical homocystinuria, or HCU, it is anticipated that the timeline for broader patient enrollment and eventual commercialization will be significantly extended. It is now expected that study enrollment will recommence in 2026. Consequently, there has been a modest adjustment in the perceived probability of success for pegtibatinase, now estimated at 60%, and a corresponding two-year postponement in the projected sales timeline.
Here are the latest investment ratings and price targets for $Travere Therapeutic (TVTX.US)$ from 4 analysts:
Note:
TipRanks, an independent third party, provides analysis data from financial analysts and calculates the Average Returns and Success Rates of the analysts' recommendations. The information presented is not an investment recommendation and is intended for informational purposes only.
Success rate is the number of the analyst's successful ratings, divided by his/her total number of ratings over the past year. A successful rating is one based on if TipRanks' virtual portfolio earned a positive return from the stock. Total average return is the average rate of return that the TipRanks' virtual portfolio has earned over the past year. These portfolios are established based on the analyst's preliminary rating and are adjusted according to the changes in the rating.
TipRanks provides a ranking of each analyst up to 5 stars, which is representative of all recommendations from the analyst. An analyst's past performance is evaluated on a scale of 1 to 5 stars, with more stars indicating better performance. The star level is determined by his/her total success rate and average return.
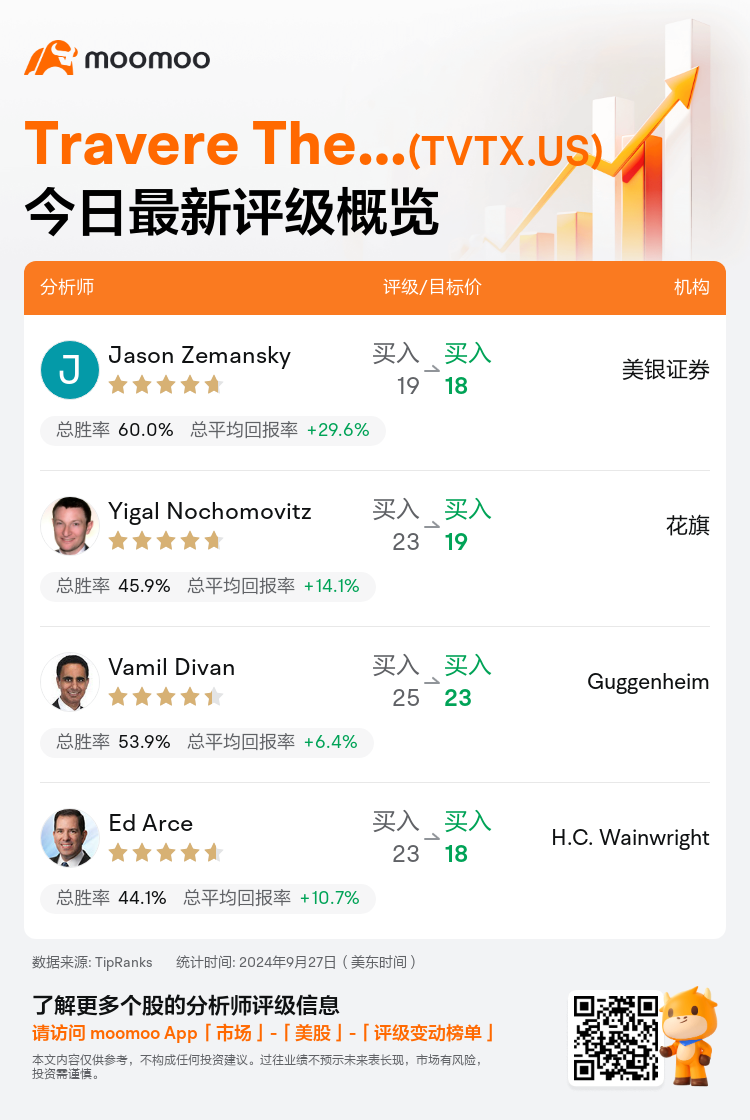
美东时间9月27日,多家华尔街大行更新了$Travere Therapeutic (TVTX.US)$的评级,目标价介于18美元至23美元。
美银证券分析师Jason Zemansky维持买入评级,并将目标价从19美元下调至18美元。
花旗分析师Yigal Nochomovitz维持买入评级,并将目标价从23美元下调至19美元。
 Guggenheim分析师Vamil Divan维持买入评级,并将目标价从25美元下调至23美元。
Guggenheim分析师Vamil Divan维持买入评级,并将目标价从25美元下调至23美元。
H.C. Wainwright分析师Ed Arce维持买入评级,并将目标价从23美元下调至18美元。
此外,综合报道,$Travere Therapeutic (TVTX.US)$近期主要分析师观点如下:
Travere Therapeutics最近宣布,出于制造方面的考虑,其聚乙二醇酶3期HARMONY研究的注册暂停。这一事态发展被视为商业投资组合的延迟,该公司预计最早可能在2026年恢复试验。尽管遇到了挫折,但该计划的价值已经微乎其微,该公司的管理层已经表示,暂停不太可能影响试验的设计和注册。因此,预计短期内对市场情绪的预期影响将是有限的。
Travere Therapeutics宣布自愿暂停其聚乙二醇酶的3期HARMONY研究的入组,原因是生产规模扩大面临的挑战。鉴于聚乙二醇酶有望将公司的产品组合扩展到Filspari以外,这一发展被视为挫折。预期的市场引入现已调整为2030财年,前提是入学将于2026年重新开始。
在Travere Therapeutics宣布自愿暂停用于治疗传统高胱氨酸尿症(HCU)的聚乙二醇酶的三期HARMONY研究之后,预计扩大患者入组和最终商业化的时间表将大大延长。现在预计研究招生将在2026年重新开始。因此,pegtibatinase的预期成功概率略有调整,目前估计为60%,预计销售时间表也相应推迟了两年。
以下为今日4位分析师对$Travere Therapeutic (TVTX.US)$的最新投资评级及目标价:
提示:
TipRanks为独立第三方,提供金融分析师的分析数据,并计算分析师推荐的平均回报率和胜率。提供的信息并非投资建议,仅供参考。本文不对评级数据和报告的完整性与准确性做出认可、声明或保证。
TipRanks提供每位分析师的星级,分析师星级代表分析师所有推荐的过往表现,通过分析师的总胜率和平均回报率综合计算得出,星星越多,则该分析师过往表现越优异,最高为5颗星。
分析师总胜率为近一年分析师的评级成功次数占总评级次数的比率。评级的成功与否,取决于TipRanks的虚拟投资组合是否从该股票中产生正回报。
总平均回报率为基于分析师的初始评级创建虚拟投资组合,并根据评级变化对组合进行调整,在近一年中该投资组合所获得的回报率。
 Guggenheim分析师Vamil Divan维持买入评级,并将目标价从25美元下调至23美元。
Guggenheim分析师Vamil Divan维持买入评级,并将目标价从25美元下调至23美元。 
 Guggenheim analyst Vamil Divan maintains with a buy rating, and adjusts the target price from $25 to $23.
Guggenheim analyst Vamil Divan maintains with a buy rating, and adjusts the target price from $25 to $23.