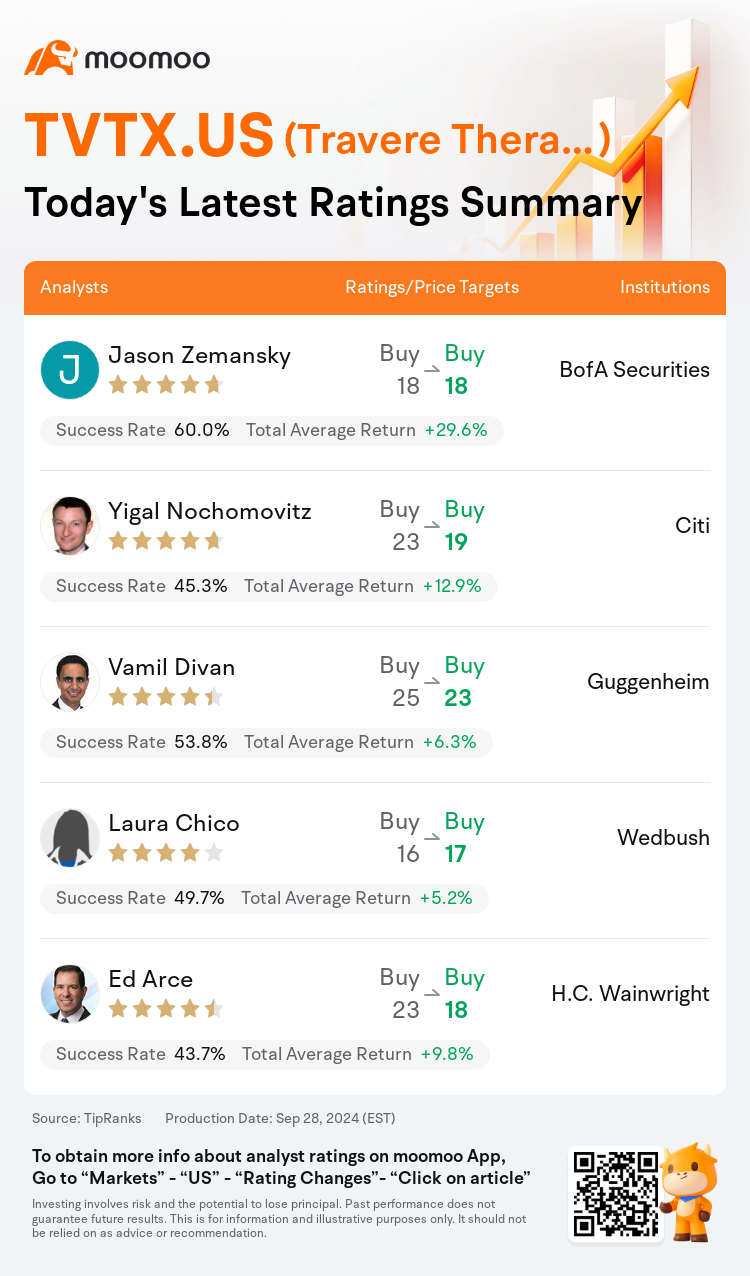
On Sep 28, major Wall Street analysts update their ratings for $Travere Therapeutic (TVTX.US)$, with price targets ranging from $17 to $23.
BofA Securities analyst Jason Zemansky maintains with a buy rating, and maintains the target price at $18.
Citi analyst Yigal Nochomovitz maintains with a buy rating, and adjusts the target price from $23 to $19.
 Guggenheim analyst Vamil Divan maintains with a buy rating, and adjusts the target price from $25 to $23.
Guggenheim analyst Vamil Divan maintains with a buy rating, and adjusts the target price from $25 to $23.
Wedbush analyst Laura Chico maintains with a buy rating, and adjusts the target price from $16 to $17.
H.C. Wainwright analyst Ed Arce maintains with a buy rating, and adjusts the target price from $23 to $18.
Furthermore, according to the comprehensive report, the opinions of $Travere Therapeutic (TVTX.US)$'s main analysts recently are as follows:
Travere Therapeutics has announced a halt in enrollment for the pegtibatinase phase 3 HARMONY study due to manufacturing challenges, which is acknowledged as a delay in the commercial portfolio. The company anticipates that the trial could restart by 2026 at the earliest. Despite the setback, the program was previously not considered highly valuable by many, and the management has highlighted that the disruptions should not affect the trial design and enrollment. Consequently, it is anticipated that the influence on near-term sentiment will be relatively limited.
Travere Therapeutics has voluntarily paused enrollment in their Phase 3 HARMONY study of pegtibatinase due to challenges encountered with manufacturing scale-up. Analysts have expressed that while the pause is unfortunate, it is noteworthy as pegtibatinase was seen as an opportunity to diversify beyond Filspari. The anticipated launch has now been adjusted to fiscal 2030, based on projections that enrollment will recommence in 2026.
Following Travere Therapeutics' announcement of a voluntary pause on the Phase 3 HARMONY study for pegtibatinase in treating classical homocystinuria, or HCU, the timeline for larger patient enrollment and eventual commercialization is now anticipated to be significantly delayed, with expectations to resume study enrollment in 2026. Consequently, the probability of success for pegtibatinase has been moderately reduced, and the forecast for sales has been deferred by two years.
Here are the latest investment ratings and price targets for $Travere Therapeutic (TVTX.US)$ from 5 analysts:
Note:
TipRanks, an independent third party, provides analysis data from financial analysts and calculates the Average Returns and Success Rates of the analysts' recommendations. The information presented is not an investment recommendation and is intended for informational purposes only.
Success rate is the number of the analyst's successful ratings, divided by his/her total number of ratings over the past year. A successful rating is one based on if TipRanks' virtual portfolio earned a positive return from the stock. Total average return is the average rate of return that the TipRanks' virtual portfolio has earned over the past year. These portfolios are established based on the analyst's preliminary rating and are adjusted according to the changes in the rating.
TipRanks provides a ranking of each analyst up to 5 stars, which is representative of all recommendations from the analyst. An analyst's past performance is evaluated on a scale of 1 to 5 stars, with more stars indicating better performance. The star level is determined by his/her total success rate and average return.
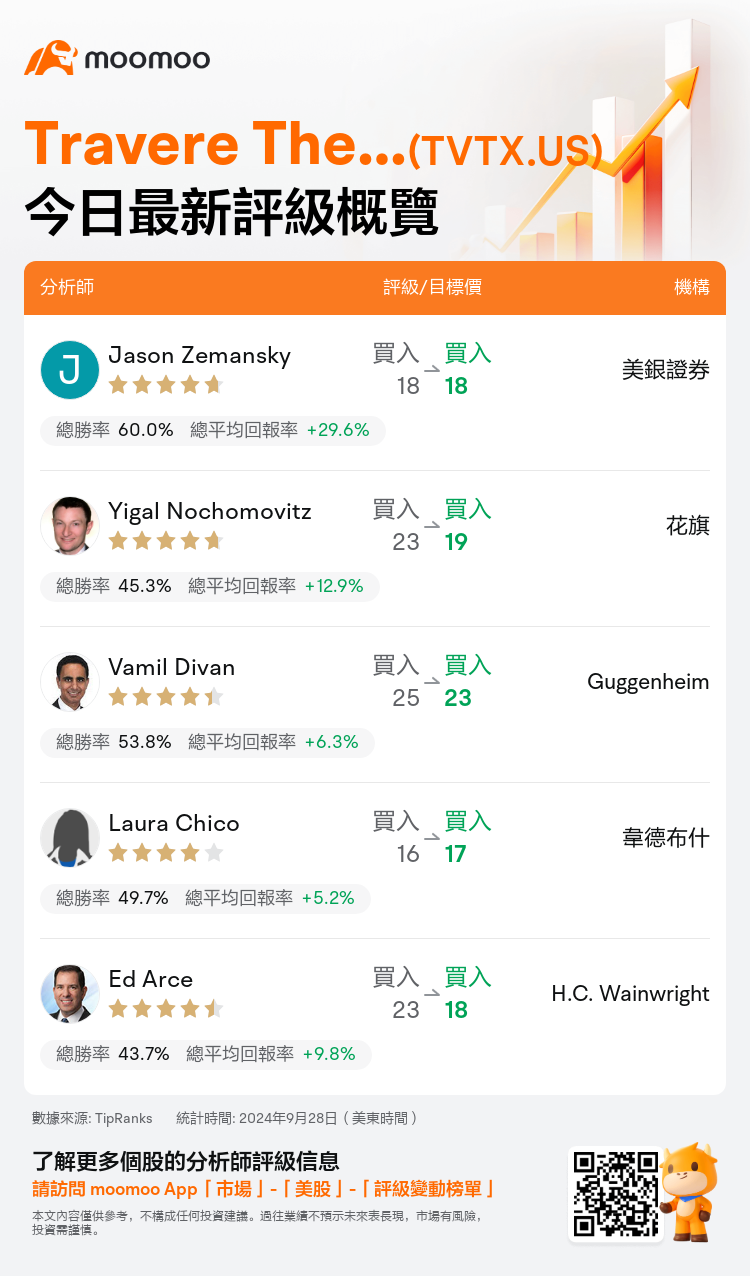
美東時間9月28日,多家華爾街大行更新了$Travere Therapeutic (TVTX.US)$的評級,目標價介於17美元至23美元。
美銀證券分析師Jason Zemansky維持買入評級,維持目標價18美元。
花旗分析師Yigal Nochomovitz維持買入評級,並將目標價從23美元下調至19美元。
 Guggenheim分析師Vamil Divan維持買入評級,並將目標價從25美元下調至23美元。
Guggenheim分析師Vamil Divan維持買入評級,並將目標價從25美元下調至23美元。
韋德布什分析師Laura Chico維持買入評級,並將目標價從16美元上調至17美元。
H.C. Wainwright分析師Ed Arce維持買入評級,並將目標價從23美元下調至18美元。
此外,綜合報道,$Travere Therapeutic (TVTX.US)$近期主要分析師觀點如下:
由於製造方面的挑戰,Travere Therapeutics宣佈暫停聚乙二醇酶3期HARMONY研究的註冊,這被認爲是商業投資組合的延遲。該公司預計,該試驗最早可能在2026年重啓。儘管遇到了挫折,但許多人此前並不認爲該計劃具有很高的價值,管理層強調中斷不應影響試驗的設計和註冊。因此,預計對短期情緒的影響將相對有限。
由於在擴大生產規模方面遇到挑戰,Travere Therapeutics自願暫停了聚乙二醇酶的3期HARMONY研究的註冊。分析師表示,儘管暫停令人遺憾,但值得注意的是,聚乙二醇酶被視爲在Filspari之外實現多元化的機會。根據預計招生將於2026年重新開始,預計的發射現已調整爲2030財年。
在Travere Therapeutics宣佈自願暫停用於治療傳統高胱氨酸尿症(HCU)的聚乙二醇酶的3期HARMONY研究之後,現在預計擴大患者入組和最終商業化的時間表將大大推遲,預計將在2026年恢復研究入組。因此,聚乙二醇酶成功的可能性已適度降低,銷售預測已推遲了兩年。
以下爲今日5位分析師對$Travere Therapeutic (TVTX.US)$的最新投資評級及目標價:
提示:
TipRanks為獨立第三方,提供金融分析師的分析數據,並計算分析師推薦的平均回報率和勝率。提供的信息並非投資建議,僅供参考。本文不對評級數據和報告的完整性與準確性做出認可、聲明或保證。
TipRanks提供每位分析師的星級,分析師星級代表分析師所有推薦的過往表現,通過分析師的總勝率和平均回報率综合計算得出,星星越多,則該分析師過往表現越優異,最高爲5颗星。
分析師總勝率為近一年分析師的評級成功次數占總評級次數的比率。評级的成功與否,取決於TipRanks的虚擬投資組合是否從該股票中產生正回報。
總平均回報率為基於分析師的初始評級創建虚擬投資組合,並根據評級變化對組合進行調整,在近一年中該投資組合所獲得的回報率。
 Guggenheim分析師Vamil Divan維持買入評級,並將目標價從25美元下調至23美元。
Guggenheim分析師Vamil Divan維持買入評級,並將目標價從25美元下調至23美元。 
 Guggenheim analyst Vamil Divan maintains with a buy rating, and adjusts the target price from $25 to $23.
Guggenheim analyst Vamil Divan maintains with a buy rating, and adjusts the target price from $25 to $23.