No Data
603392 Beijing Wantai Biological Pharmacy Enterprise
- 73.34
- -3.68-4.78%
- 5D
- Daily
- Weekly
- Monthly
- 1Q
- 1Y
Trade Overview
Capital Trend
No Data
News
Beijing Wantai Biological Pharmacy Enterprise (603392.SH): The company has started early market research for its nine-valent HPV vaccine in multiple countries overseas.
On September 9th, Beijing Wantai Biological Pharmacy Enterprise (603392.SH) stated on the interactive platform that the company has initiated early market research for its nine-valent HPV vaccine in multiple countries overseas. According to regulatory requirements, the registration and access application in foreign countries must be based on domestic registration approval.
Beijing Wantai Biological Pharmacy Enterprise (603392.SH): has accumulated repurchased 0.2271% of shares.
On September 2, Gelonhui reported that as of August 30, 2024, Beijing Wantai Biological Pharmacy Enterprise (603392.SH) has accumulated repurchased 2,880,385 shares through centralized competitive bidding trading, accounting for 0.2271% of the total share capital of the company. The highest repurchase price was 74.48 yuan/share, the lowest price was 45.55 yuan/share, and the total amount paid was RMB 186,125,317.98 (excluding trading commission and other trading expenses).
Beijing Wantai Biological Pharmacy Enterprise 2024 Semi-Annual Report.
Summary of Beijing Wantai Biological Pharmacy Enterprise's 2024 half-year report
Behind the "cliff-like" decline in performance of Beijing Wantai Biological: vaccine business revenue decreased by more than 80% | interpretations
①The main reason for the significant decline in the performance of beijing wantai biological pharmacy enterprise's H1 is the decrease in sales revenue of the bivalent HPV vaccine. ②The revenue of the company's diagnostic division exceeded the vaccine division, while overseas revenue saw a certain year-on-year growth. ③The production conditions for the commercialization of the nonavalent HPV vaccine of the company are in place, and the listing application is progressing according to plan.
Beijing Wantai Biological Pharmacy Enterprise (603392.SH): the nine-valent HPV vaccine is expected to be included in the priority review program.
On August 12th, Gelunhui reported that Beijing Wantai Biological Pharmacy Enterprise (603392.SH) announced that its wholly-owned subsidiary, Xiamen Wantai Canghai Biotechnology Co., Ltd. collaborated with Xiamen University to research and develop a nine-valent vaccine (Escherichia coli) for human papillomavirus type 6/11/16/18/31/33/45/52/58 (referred to as 'nine-valent HPV vaccine'), which is on the priority list of drugs for review and approval by the National Medical Products Administration's drug evaluation center, with a public notice period of 7 days.
Comments
Securities: $CITIC (600030.SH)$ $East Money Information (300059.SZ)$
Insurance: $China Life Insurance (601628.SH)$ $Ping An Insurance (601318.SH)$
Auto Parts: $Fuyao Glass Industry Group (600660.SH)$ $Weichai Power (000338.SZ)$ $Ningbo Tuopu Group (601689.SH)$
Cars: $BYD Company Limited (002594.SZ)$
Software: $Beijing Kingsoft Office Software, Inc (688111.SH)$ $Hithink RoyalFlush Information Network (300033.SZ)$ $Iflytek Co.,ltd. (002230.SZ)$
���������...
Wantai Biolog 万泰生物 120MIN Chart - bullish Adam & Eve breakout, possible to hit ¥80
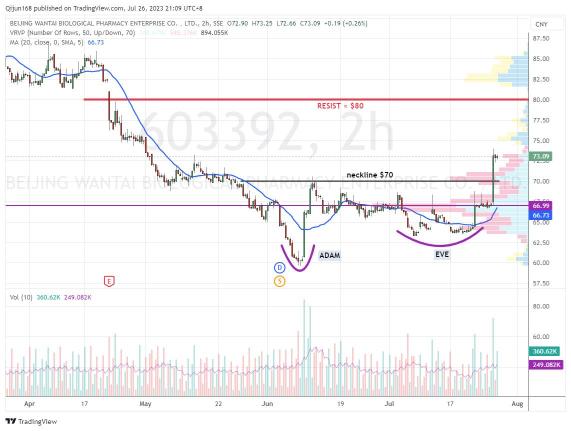
Analysis
Price Target
No Data
No Data
