No Data
BGNE240920C115000
- 0.00
- 0.000.00%
- 5D
- Daily
No Data
News
Merck, Bristol Myers Cancer Drugs Included in September FDA Advisory Panel Meeting
Beigene (06160) class 1 new drug BGB-43395 tablet approved for clinical use in breast cancer and other solid tumors.
On August 22nd, the official website of China National Medical Products Administration (NMPA) Drug Evaluation Center (CDE) announced that Beigene (06160) has received clinical approval for the first class of new drug BGB-43395 tablets. It is intended to be used for monotherapy or in combination with fluorouracil and vinorelbine or other drugs for the treatment of advanced or metastatic HR+/HER2- breast cancer and other solid tumors.
HKEX abnormal movement| BeiGene (06160) rose nearly 3% in the morning, and many innovative drug companies welcomed their mid-year reports with joy. The company's BTK drug market share continues to expand in the United States.
Beigene (06160) rose nearly 3% in the morning. As of press time, it rose 2.82% to HKD 120.5, with a turnover of HKD 90.0317 million.
BeiGene Is Maintained at Overweight by JP Morgan
BeiGene Analyst Ratings
J.P. Morgan Maintains BeiGene(BGNE.US) With Buy Rating, Raises Target Price to $200
Comments
⇨ Imfinzi + chemotherapy
‣ Neoadjuvant treatment NSCLC
‣ AdCom: 7/25/24 (sBLA)
$BeiGene (BGNE.US)$ : 🤔
⇨ TEVIMBRA (tislelizumab)
‣ Esophageal squamous-cell carcinomas (1L)
‣ PDUFA: July 2024 (sBLA)
$Phathom Pharmaceuticals (PHAT.US)$ : 🤔
⇨ VOQUEZNA® (vonoprazan)
‣ Non-Erosive GERD
‣ PDUFA: 7/19/24 (NDA)
$AstraZeneca (AZN.US)$ : AdCom 🤔
⇨ Imfinzi + chemotherapy
‣ Neoadjuvant treatment NSCLC
‣ AdCom: 7/25/24(sBLA)
$BeiGene (BGNE.US)$ : 🤔
⇨ TEVIMBRA (tislelizumab)
‣ Esophageal squamous-cell carcinomas
‣ PDUFA: July 2024 (BLA)

$Arcutis Biotherapeutics (ARQT.US)$ : pending decision 🤔
⇨ ARQ-151 topical roflumilast cream
‣ Atopic Dermatitis (aged 6 & older children)
‣ PDUFA date: 7/7/24 (sNDA)
Upcoming PDUFAs & AdCom in July: 📅👇
$Phathom Pharmaceuticals (PHAT.US)$ : 🤔
⇨ VOQUEZNA® (vonoprazan)
‣ Non-Erosive GERD
‣ PDUFA date: 7/19/24 (NDA)
$AstraZeneca (AZN.US)$ : AdCom 🤔
⇨ Imfinzi + chemotherapy
‣ Neoadjuvant treatment NSCLC
‣ AdCom date: 7/25/24(sBLA)
$BeiGene (BGNE.US)$ :...

$Arcutis Biotherapeutics (ARQT.US)$ : ARQ-151 topical roflumilast cream
$Phathom Pharmaceuticals (PHAT.US)$ : VOQUEZNA® (vonoprazan)
$AstraZeneca (AZN.US)$ : Imfinzi + chemotherapy - AdCom
$BeiGene (BGNE.US)$ : TEVIMBRA (tislelizumab)

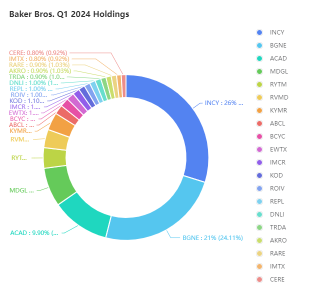
» Holdings in Top 20: 86.71%
» Qtr over Qtr Change**: -9.29%
Top Holdings:
$Incyte (INCY.US)$
$BeiGene (BGNE.US)$
$ACADIA Pharmaceuticals (ACAD.US)$
$Madrigal Pharmaceuticals (MDGL.US)$
$Rhythm Pharmaceuticals (RYTM.US)$
$Revolution Medicines (RVMD.US)$
$Kymera Therapeutics (KYMR.US)$
$AbCellera Biologics (ABCL.US)$
$Bicycle Therapeutics (BCYC.US)$
$Edgewise Therapeutics (EWTX.US)$
$Immunocore (IMCR.US)$
$Kodiak Sciences (KOD.US)$
$Roivant Sciences (ROIV.US)$
���������...