No Data
US Stock MarketDetailed Quotes
LTRN Lantern Pharma
- 3.440
- +0.050+1.47%
Close Oct 18 16:00 ET
- 3.440
- 0.0000.00%
Post 20:01 ET
37.03MMarket Cap-2084P/E (TTM)
3.500High3.350Low17.11KVolume3.350Open3.390Pre Close58.78KTurnover0.22%Turnover RatioLossP/E (Static)10.76MShares11.99052wk High1.20P/B27.33MFloat Cap2.38052wk Low--Dividend TTM7.94MShs Float24.840Historical High--Div YieldTTM4.43%Amplitude2.380Historical Low3.434Avg Price1Lot Size
Full Hours
- 5D
- Daily
- Weekly
- Monthly
- 1Q
- 1Y
Trade Overview
Unit: --
Capital Trend
IntradayDayWeekMonth
No Data
News
Express News | Lantern Pharma Inc - Phase 1B/2a Trial for Recurrent GBM to Start Late 2024
Lantern Pharma's Investigational Drug-Candidate, LP-184, Receives Fast-Track Designation in Glioblastoma From the FDA
Express News | Lantern Pharma’s Investigational Drug-Candidate, LP-184, Receives Fast-Track Designation in Glioblastoma From the FDA
Lantern Pharma Inc (LTRN) Q2 2024 Earnings Call Highlights: Promising Clinical Trials and ...
Lantern Appoints Piers Dixon as Head of Sales
Lantern Pharma Says FDA Oks Rare Pediatric Disease Designations In MRT, RMS, And Hepatoblastoma
Comments
$China Pharma (CPHI.US)$ $Bright Minds Biosciences (DRUG.US)$ $Lantern Pharma (LTRN.US)$ $Cytosorbents (CTSO.US)$ $Intensity Therapeutics (INTS.US)$ $Multi Ways (MWG.US)$ $Salarius Pharmaceuticals (SLRX.US)$ $X3 Holdings (XTKG.US)$ $Protagenic Therapeutics (PTIX.US)$ $GRI Bio (GRI.US)$ $Paranovus Entertainment Technology (PAVS.US)$ $VSee Health (VSEE.US)$
📊⚡️📊
📊⚡️📊
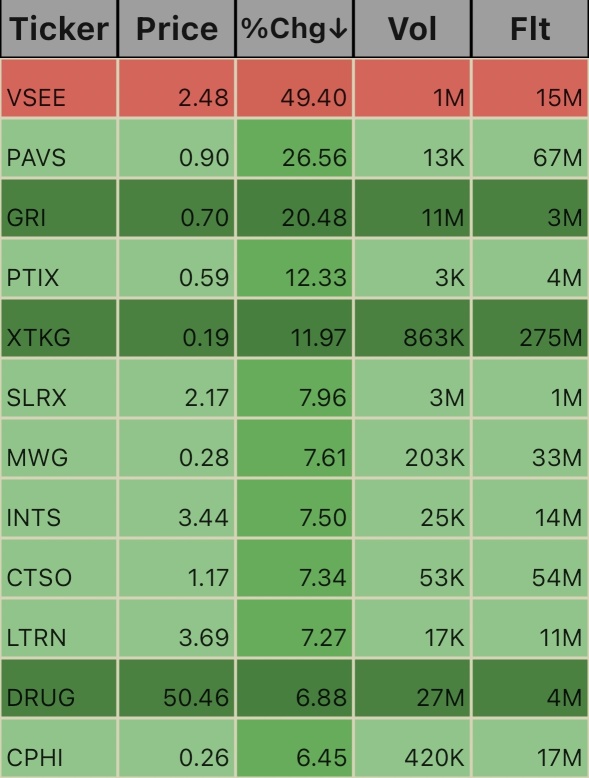
2
1
$Lantern Pharma (LTRN.US)$ Lantern Pharma's Investigational Drug-Candidate, LP-184, Receives Fast-Track Designation in Glioblastoma from the FDA
6 MINUTES AGO, 9:07 AM EDT
VIA BUSINESSWIRE
6 MINUTES AGO, 9:07 AM EDT
VIA BUSINESSWIRE
$Hall Of Fame Resort & Entertainment (HOFV.US)$ $Vera Bradley (VRA.US)$ $Lantern Pharma (LTRN.US)$ $Passage Bio (PASG.US)$ $Sadot (SDOT.US)$ $EDC Publishing (EDUC.US)$ $SHL Telemedicine (SHLT.US)$ $Altamira Therapeutics (CYTO.US)$ $Indaptus Therapeutics (INDP.US)$ $QT Imaging (QTI.US)$ $MediaCo (MDIA.US)$ $Edesa Biotech (EDSA.US)$ $Expion360 (XPON.US)$ $Tevogen Bio Holdings (TVGN.US)$
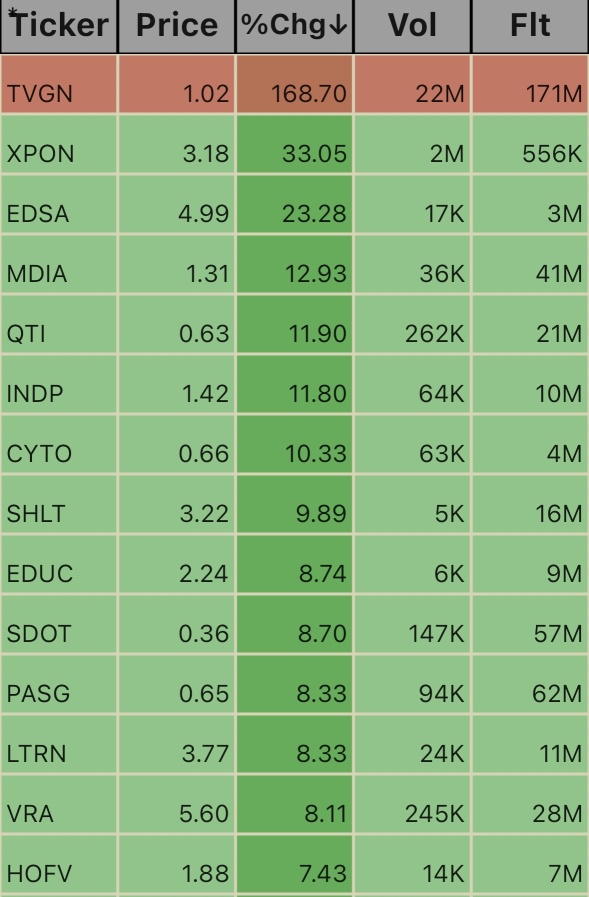
1
$Lantern Pharma (LTRN.US)$
Lantern Pharma Announces Three U.S. FDA Rare Pediatric Disease Designations Granted to LP-184 in Multiple Ultra Rare Children's Cancers
• Lantern Pharma was granted the rare pediatric disease designation (RPDD) for drug-candidate, LP-184, in three cancer indications: Malignant Rhabdoid Tumors, Rhabdomyosarcoma, and Hepatoblastoma.
• This brings the total number of RPDDs for LP-184 to 4, including one previously granted for ATRT (Atypical Teratoid Rhabdoid Tumors)....
Lantern Pharma Announces Three U.S. FDA Rare Pediatric Disease Designations Granted to LP-184 in Multiple Ultra Rare Children's Cancers
• Lantern Pharma was granted the rare pediatric disease designation (RPDD) for drug-candidate, LP-184, in three cancer indications: Malignant Rhabdoid Tumors, Rhabdomyosarcoma, and Hepatoblastoma.
• This brings the total number of RPDDs for LP-184 to 4, including one previously granted for ATRT (Atypical Teratoid Rhabdoid Tumors)....
$Lantern Pharma (LTRN.US)$ This possible play is purely chart driven! Thats the reason im sharing. So those that asked. Here is a example of some of what i look for. This is what brought my attention here🤔🤫
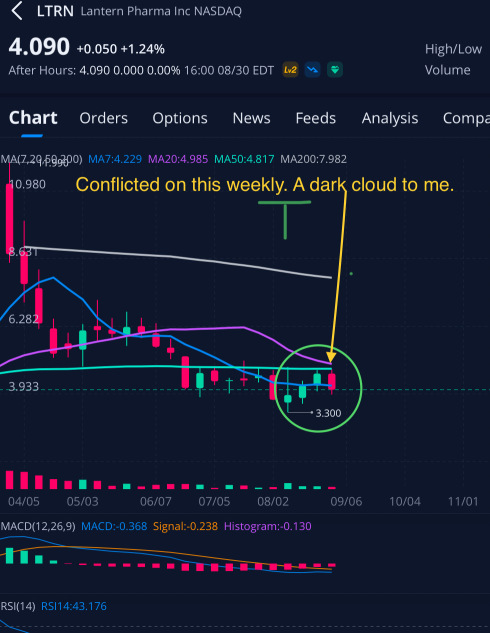
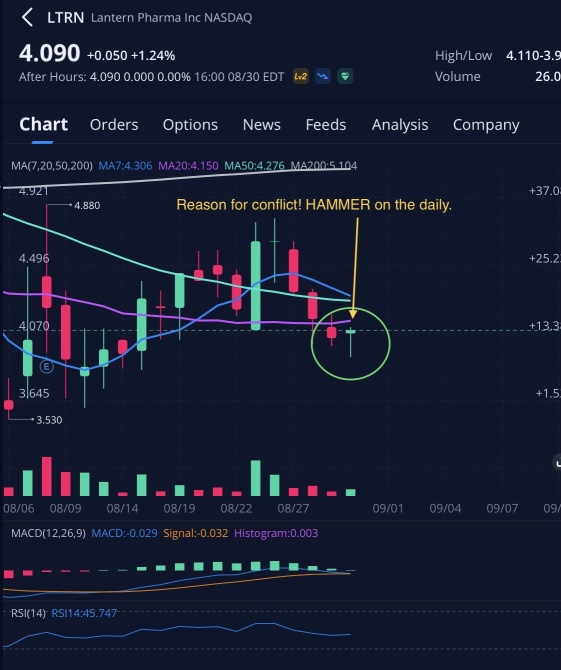
4
1
Read more
Analysis
Price Target
No Data
Heat List
Overall
Symbol
Latest Price
% Chg
No Data

jimmy the trader : $China Pharma (CPHI.US)$ broths