No Data
US Stock MarketDetailed Quotes
BMY Bristol-Myers Squibb
- 48.500
- -0.200-0.41%
Close Sep 9 16:00 ET
- 48.590
- +0.090+0.19%
Post 19:58 ET
98.33BMarket Cap-14923P/E (TTM)
49.125High47.550Low9.43MVolume48.720Open48.700Pre Close458.47MTurnover0.47%Turnover Ratio12.56P/E (Static)2.03BShares56.65652wk High5.78P/B98.15BFloat Cap39.03652wk Low2.37Dividend TTM2.02BShs Float75.683Historical High4.89%Div YieldTTM3.23%Amplitude8.383Historical Low48.601Avg Price1Lot Size
Full Hours
- 5D
- Daily
- Weekly
- Monthly
- 1Q
- 1Y
Trade Overview
Unit: --
Capital Trend
IntradayDayWeekMonth
No Data
News
Enliven Therapeutics' Leukemia Candidate Could Challenge Pfizer, AstraZeneca In Broader Target Market: Analyst
Bristol Myers Squibb to Present Data at ESMO Demonstrating Ongoing Leadership in Immuno-Oncology and Progression of Assets From Its Differentiated Research Platforms
Bristol-Myers Squibb (BMY): Hold Rating Justified by Mixed Growth Prospects and Pipeline Catalysts
Zenas BioPharma, Bicara Therapeutics Sets IPO Price Ranges as Summer Lull Ends
Bristol Myers Squibb Employees to Relay Nearly 3,000 Miles Cross-Country Aspiring to Raise Over $1 Million for the V Foundation for Cancer Research
Zenas BioPharma Sets Price Range for $200 Million Initial Public Offering
Comments
🚀 Buckle up for upcoming PDUFAs in September! 👇
$Travere Therapeutic (TVTX.US)$ : FILSPARI (Sparsentan)
$Avadel Pharmaceuticals (AVDL.US)$ : LUMRYZ (FT218)
$Iterum Therapeutics (ITRM.US)$ : Sulopenem - AdCom 🗓️
$Sanofi (SNY.US)$ & $Regeneron Pharmaceuticals (REGN.US)$ : Dupixent
$Vanda Pharmaceuticals (VNDA.US)$ : Tradipitant
$Zevra Therapeutics (ZVRA.US)$ : Arimoclomol
$Heron Therapeutics (HRTX.US)$ : ZYNRELEF® Vial Access Needle
$Merck & Co (MRK.US)$ : KEYTRUDA+ chemotherapy
$Bristol-Myers Squibb (BMY.US)$ : KarXT (...
$Travere Therapeutic (TVTX.US)$ : FILSPARI (Sparsentan)
$Avadel Pharmaceuticals (AVDL.US)$ : LUMRYZ (FT218)
$Iterum Therapeutics (ITRM.US)$ : Sulopenem - AdCom 🗓️
$Sanofi (SNY.US)$ & $Regeneron Pharmaceuticals (REGN.US)$ : Dupixent
$Vanda Pharmaceuticals (VNDA.US)$ : Tradipitant
$Zevra Therapeutics (ZVRA.US)$ : Arimoclomol
$Heron Therapeutics (HRTX.US)$ : ZYNRELEF® Vial Access Needle
$Merck & Co (MRK.US)$ : KEYTRUDA+ chemotherapy
$Bristol-Myers Squibb (BMY.US)$ : KarXT (...

9
4
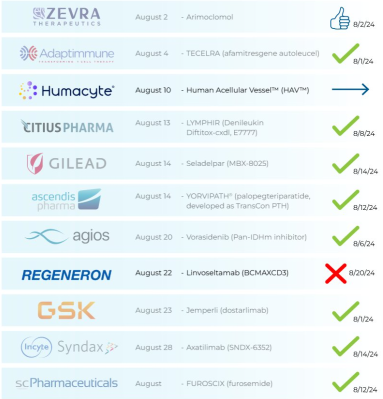
🚨 This Week’s PDUFAs:
$Regeneron Pharmaceuticals (REGN.US)$ : 🤔
⇨ Linvoseltamab
‣ Multiple myeloma
‣ PDUFA: 8/22/24 (BLA)
🗓️ Last Week’s PDUFAs:
$Gilead Sciences (GILD.US)$ : Approved 8/14 🎉
⇨ Livdelzi (Seladelpar)
‣ Primary biliary cholangitis (PBC)
‣ PDUFA: 8/14/24 (NDA)
$Ascendis Pharma A/S (ASND.US)$ : Approved 8/12 🎉
⇨ YORVIPATH (palopegteriparatide)
‣ Hypoparathyroidism
‣ PDUFA: 8/14/24 (NDA)
$Syndax Pharmaceuticals (SNDX.US)$ & $Incyte (INCY.US)$ : Approved 8/14 🎉
⇨ Nikt...
$Regeneron Pharmaceuticals (REGN.US)$ : 🤔
⇨ Linvoseltamab
‣ Multiple myeloma
‣ PDUFA: 8/22/24 (BLA)
🗓️ Last Week’s PDUFAs:
$Gilead Sciences (GILD.US)$ : Approved 8/14 🎉
⇨ Livdelzi (Seladelpar)
‣ Primary biliary cholangitis (PBC)
‣ PDUFA: 8/14/24 (NDA)
$Ascendis Pharma A/S (ASND.US)$ : Approved 8/12 🎉
⇨ YORVIPATH (palopegteriparatide)
‣ Hypoparathyroidism
‣ PDUFA: 8/14/24 (NDA)
$Syndax Pharmaceuticals (SNDX.US)$ & $Incyte (INCY.US)$ : Approved 8/14 🎉
⇨ Nikt...
8
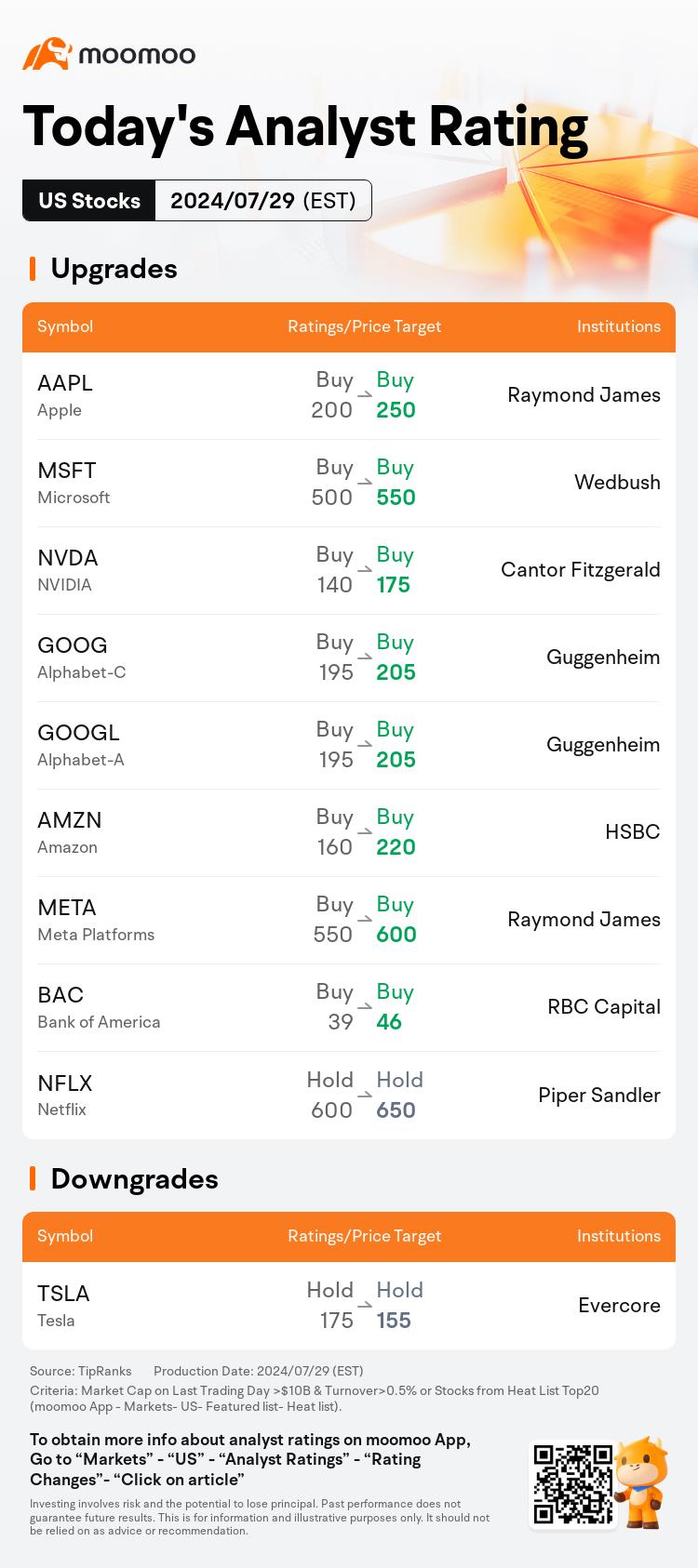
Morning Movers
Gapping up
$Royal Philips (PHG.US)$ experienced a significant boost of 10.6% after the company reported robust second-quarter earnings, attributing much of their success to cost-saving measures, including workforce reductions, and a substantial insurance payout related to liability claims.
$Tesla (TSLA.US)$ 's shares advanced by 1.9% following an endorsement by Morgan Stanley analyst Adam Jonas, naming it the top ...
Gapping up
$Royal Philips (PHG.US)$ experienced a significant boost of 10.6% after the company reported robust second-quarter earnings, attributing much of their success to cost-saving measures, including workforce reductions, and a substantial insurance payout related to liability claims.
$Tesla (TSLA.US)$ 's shares advanced by 1.9% following an endorsement by Morgan Stanley analyst Adam Jonas, naming it the top ...
33
8
$Bristol-Myers Squibb (BMY.US)$ It's about DAYUM time this stock showed some life! Hopefully it won't give it all back!
1
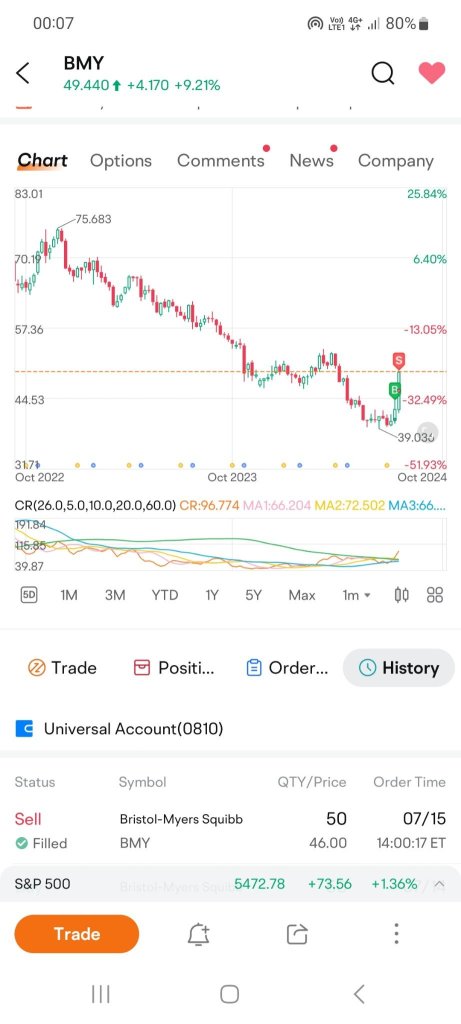
Sold this stock today for a marginal gain to free up some cash to get a better quality stock. When I started to build my portfolio on 9 June, I made a lot of rookie mistakes. while this stock is not one of those, the price was right to let it go for now. I will be circling back to this stock because it is undervalued and has growth potential.
Do you think this is a right move?
Do you think this is a right move?
2
5
Read more
Analysis
Price Target
No Data
Heat List
Overall
Symbol
Latest Price
% Chg


RDK79 : Sorry, what’s a pdufa?
Xyno RDK79 : Prescription Drug User Fee Act, means FDA approval or rejection for a drug to go to market. One of the most important events to monitor if you are into pharma companies stock.
RDK79 Xyno : Thank you!
Tonyco : What site is that?